| Active substance |
Tenofovir |
| Also known as |
TDF |
| Blood pressure |
No significant effect |
| Chemical name |
9-[(R)-2-[[Bis[[(isopropoxycarbonyl)oxy]methoxy]phosphinyl]methoxy]propyl]adenine fumarate (1:1) |
| Dosage (medical) |
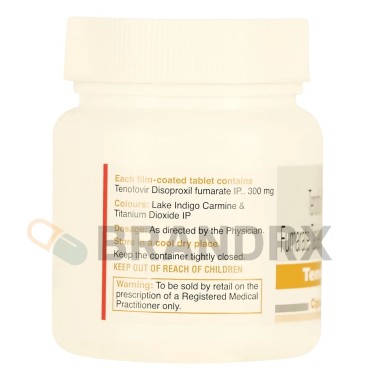
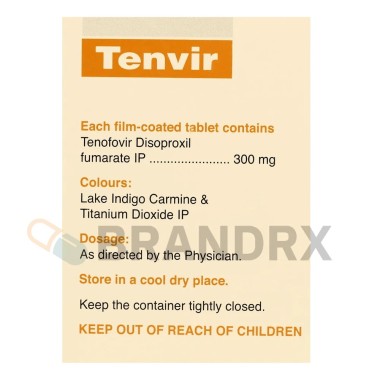
300 mg once daily |
| Dosage (sports) |
Not applicable |
| Effects |
Reduces the viral load in HIV-infected individuals, improves immune function |
| Formula |
C19H30N5O10P В· C4H4O4 |
| Half-life |
Approximately 17 hours |
| Hepatotoxicity |
Low risk |
| Lab Test |
Monitoring of renal function and bone density |
| Main action |
Antiretroviral, inhibits HIV reverse transcriptase |
| Side effects |
Nausea, vomiting, diarrhea, headache, dizziness, fatigue, renal impairment, bone loss |
| Storage conditions |
Store at 20В°C to 25В°C (68В°F to 77В°F), excursions permitted between 15В°C to 30В°C (59В°F to 86В°F) |
| Substance class |
Nucleotide reverse transcriptase inhibitor (NtRTI) |
| Trade name |
Viread |
| Use in sports |
None |
| Water Retention |
No significant effect |
| Manufacturer |
Cipla Ltd. |
| Packing |
30 tabs/bottle |